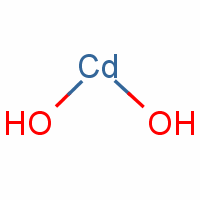
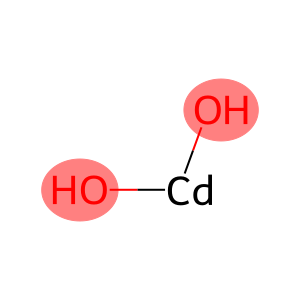
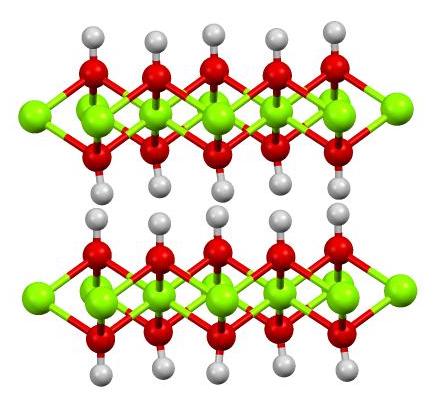
Cd/Cd(OH)2 Nanosheets Enhancing the Electrocatalytic Activity of CO2 Reduction to CO - Jia - 2023 - Chemistry – A European Journal - Wiley Online Library

Remarkable positive effect of Cd(OH)2 on CdS semiconductor for visible-light photocatalytic H2 production - ScienceDirect

Solubility of `Cd(OH)_(2)` in pure water is `1.84xx10^(-5)\"mole\"//L` Calculate its solubility in - YouTube

Equilibrium d 36. The solubility of Cd(OH), pH = 13 25°C is (Ksp of Cd(OH)2 = 2.5 10-14) (1) 2.7 * 10-13 (2) 1.09 10-10 (3) 2.5 * 10-12 (4) 1.6 10-14
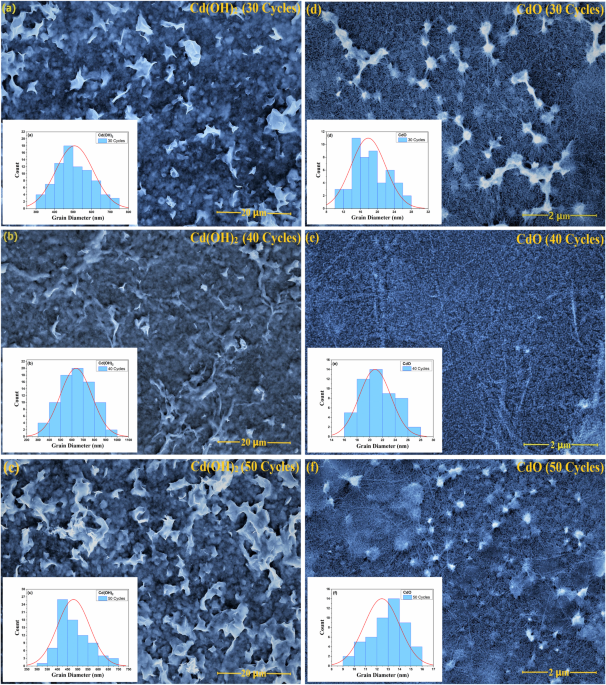
CO gas sensing properties of Cd(OH)2/CdO thin films synthesized by SILAR method | Journal of Sol-Gel Science and Technology
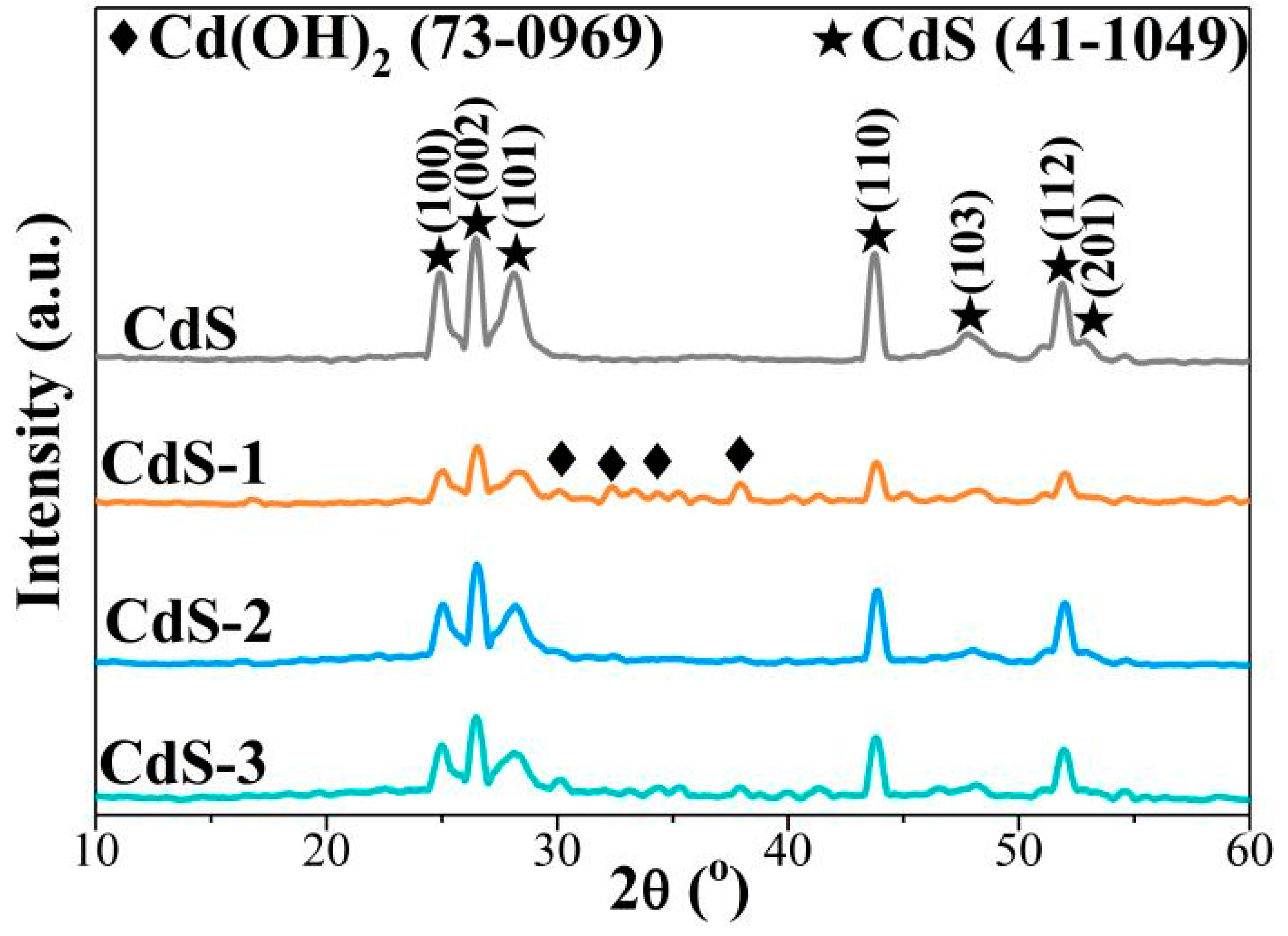
Nanomaterials | Free Full-Text | In Situ Fabrication of CdS/Cd(OH)2 for Effective Visible Light-Driven Photocatalysis

The molar solubility iof Cd(OH)_(2) is 1.84xx10^(-5)M in water. The expected solubiliyt of Cd(OH... - YouTube

SOLVED: What is the pH for a saturated solution of Cd(OH)2? The Ksp for Cd( OH)2 is 5.9 x 10^–15. pH =
The molar solubility of Cd(OH)2 is 1.84 × 10^–5 M in water. The expected solubility of Cd(OH)2 in a buffer solution of pH = 12 is : - Sarthaks eConnect | Largest Online Education Community
Poly want an ion?. This is the formula for cadmium hydroxide: 1. CdOH 2. CdOH 2 3. Cd(OH) 2 4. CdH 2 5. Cd(OH 2 ) 2 6. Cd 2 (OH) 2 7. Cd 2 H. - ppt download

CO gas sensing properties of Cd(OH)2/CdO thin films synthesized by SILAR method | Journal of Sol-Gel Science and Technology